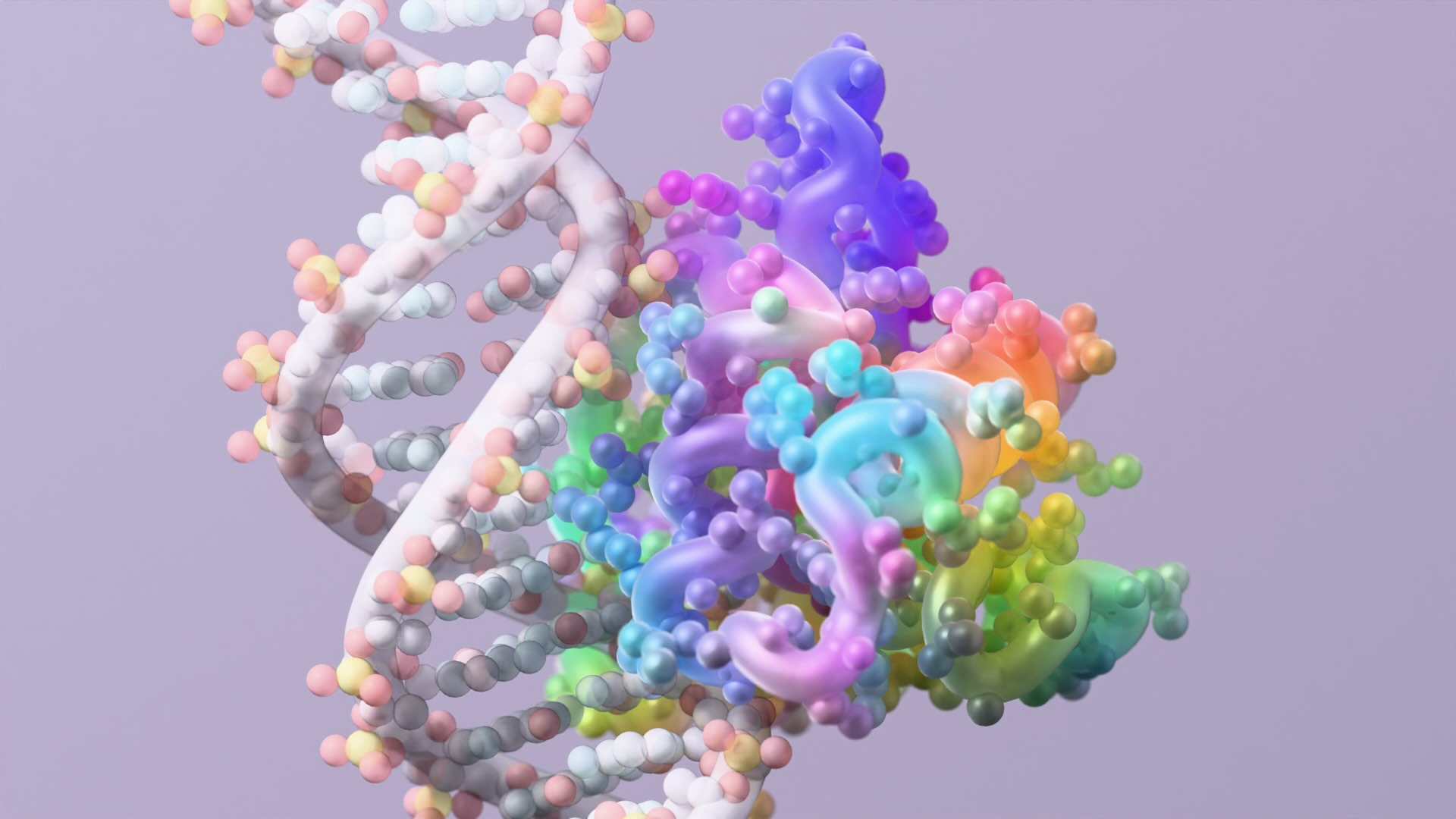
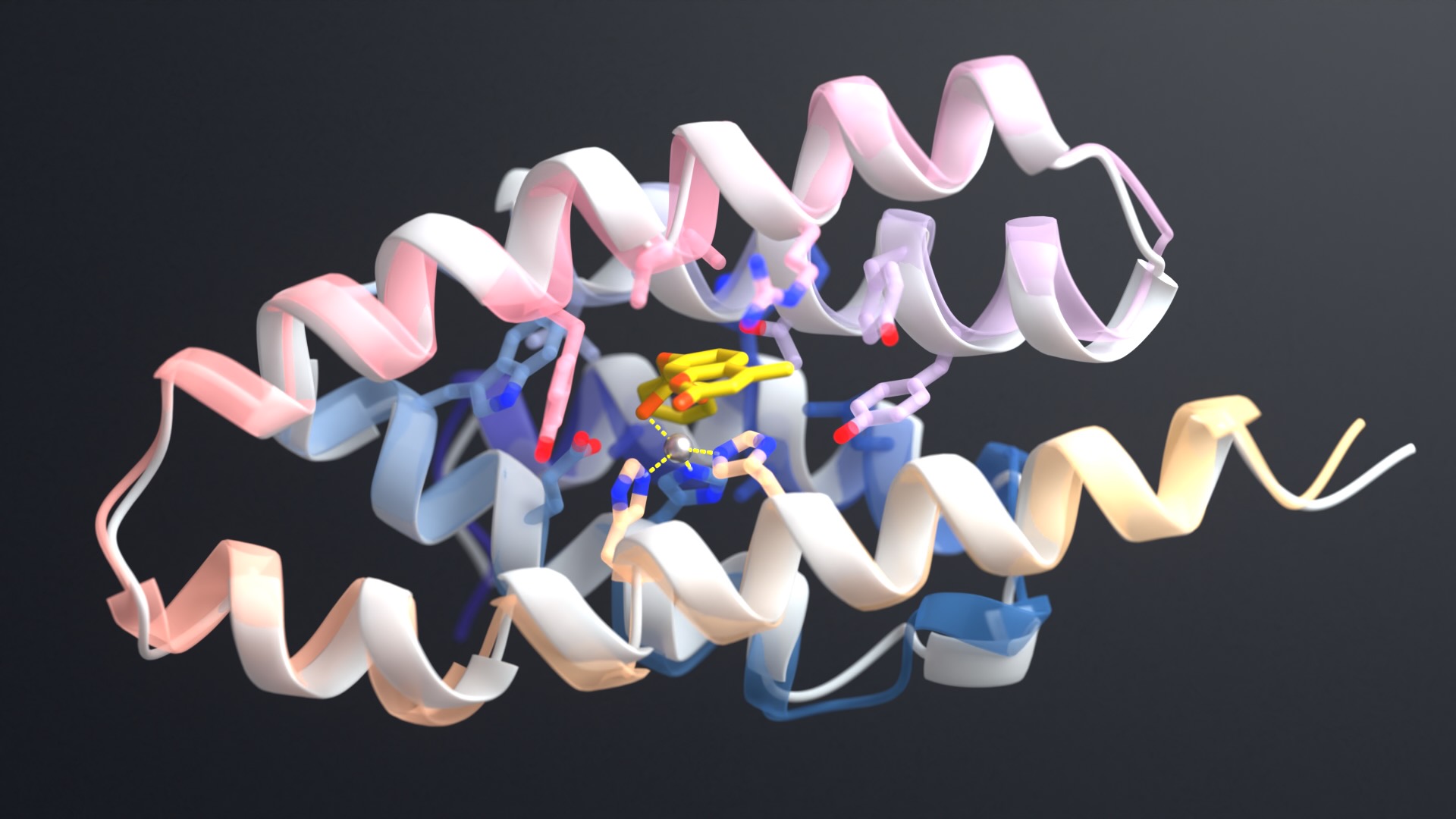
Today we report in Nature the design of proteins that recognize and bind to the so-called “intrinsically disordered regions” of proteins and peptides. The body produces such disordered molecules naturally, but many have been linked to health disorders, including myeloma and other cancers.
“Disordered proteins play important roles in biology. By designing new proteins that latch onto them, we may finally be able to diagnose and treat many burdensome diseases,” said senior author David Baker, director of the Institute for Protein Design
Traditionally, scientists who seek to develop new drugs focus on targets that have a defined structure. Antibody therapies for COVID-19, for example, work by recognizing the tell-tale shape of the Spike protein that protrudes from the surface of the coronavirus.
But how can a drug recognize a shape-shifting molecule in the body?
“To create proteins that bind to intrinsically disordered targets, we chose to go after something that’s common to all peptides and proteins: their chemical backbone.”
— Kejia Wu, co-lead author and Baker Lab graduate student

Proteins, and their shorter cousins peptides, are chains of chemicals called amino acids. The body uses an alphabet of 20 amino acids to construct its roughly 20,000 unique proteins. But every amino acid inside every protein and peptide chain shares certain chemical features. Whether those chains are structured or unstructured, this common chemical backbone remains.
Wu, together with co-lead author and recent Baker Lab postdoctoral scholar Hua Bai, devised a new protein design strategy that focuses on the inherent chemical features found in the backbone of every intrinsically disordered protein and peptide. Using computational protein design, they were able to create new proteins that bind to disordered peptides and proteins.

When tested in the lab, the best of their new proteins latched on to a disordered target for over 2,000 seconds. In one experiment, the team also showed that it was possible to design proteins that recognize and bind to a specific human protein that is known to contain an intrinsically disordered region. That target protein, called ZFC3H1, may be a biomarker for cancer.
The team also show that their new method can be used to recognize specific linear peptide sequences, which could have great utility in medical and proteomic research, including for peptide sequencing.
Read the manuscript:
Wu K., Bai H., et al. De novo design of modular peptide-binding proteins by superhelical matching. Nature, 2023